 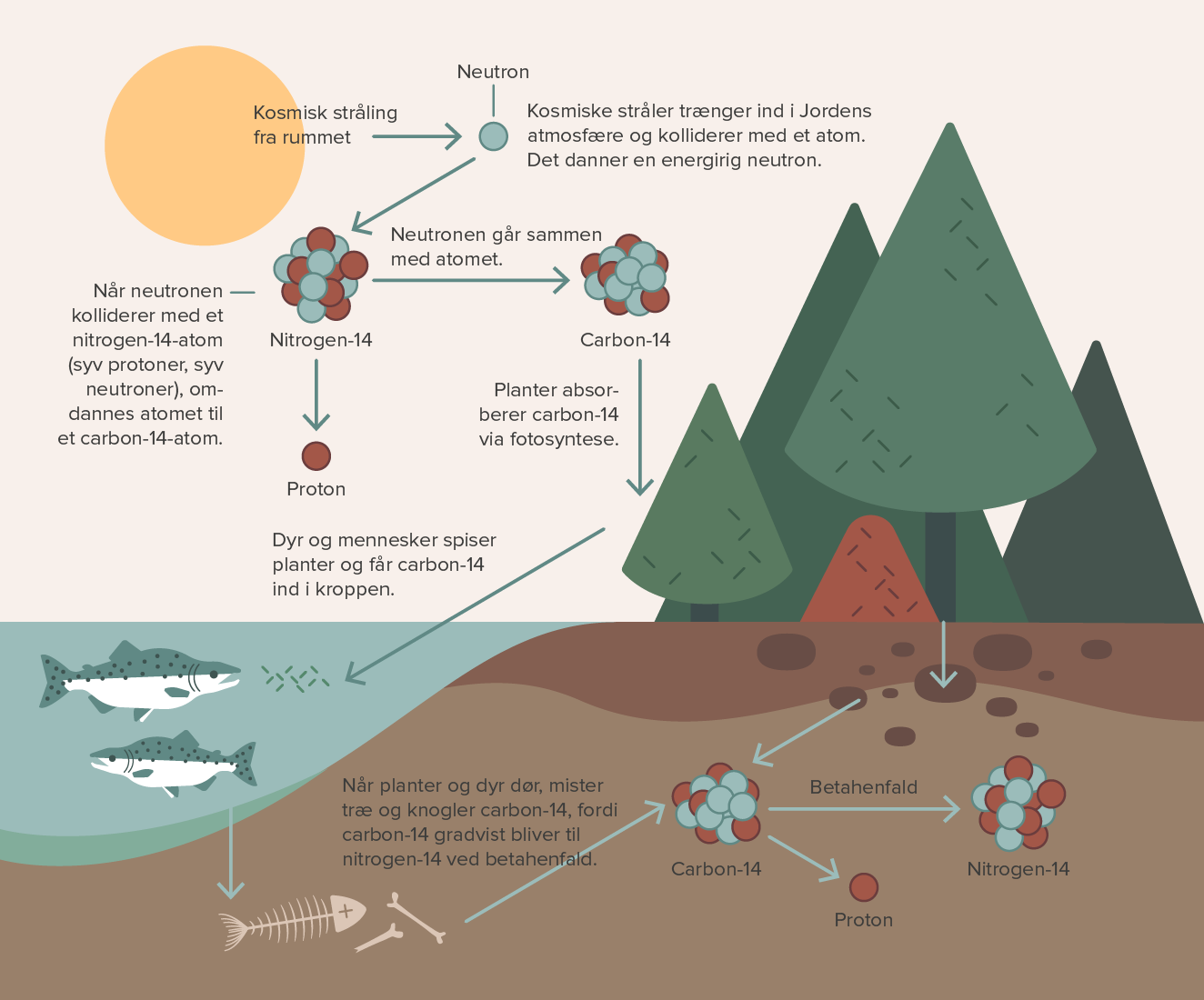
See also Fossils and fossilization GeochemistryĬarbon dating ( radiocarbon dating) A method of estimating the ages of archaeological specimens of biological origin. A newer method of radioactive tracing involves the use of a new clock, based on the radioactive decay of 235uranium to 231protactinium. 
Using the cyclotron, carbon –14 dating could be used for objects as old as 100,000 years, while samples containing radioactive beryllium could be dated as far back as 10 –30 million years. They also improved the equipment used to detect these elements, and in 1939, scientists first used a cyclotron particle accelerator as a mass spectrometer. They developed the uranium-thorium method, the potassium-argon method, and the rubidium-strontium method, all of which are based on the transformation of one element into another. Starting where Boltwood and Libby left off, scientists began to search for other long-lived isotopes. Still, even with the help of laboratories worldwide, radiocarbon dating was only accurate up to 70,000 years old, since objects older than this contained far too little carbon –14 for the equipment to detect. Using the carbon –14 method, scientists determined the ages of artifacts from many ancient civilizations. Libby's method, called radiocarbon or carbon –14 dating, gave new impetus to the science of radioactive dating. 
He continued his research and, through improvements in his equipment and procedures, was eventually able to determine the age of an object up to 50,000 years old with a precision of plus-or-minus 10%. He found that his methods, while not as accurate as he had hoped, were fairly reliable. Libby began testing his carbon –14 dating procedure by dating objects whose ages were already known, such as samples from Egyptian tombs. 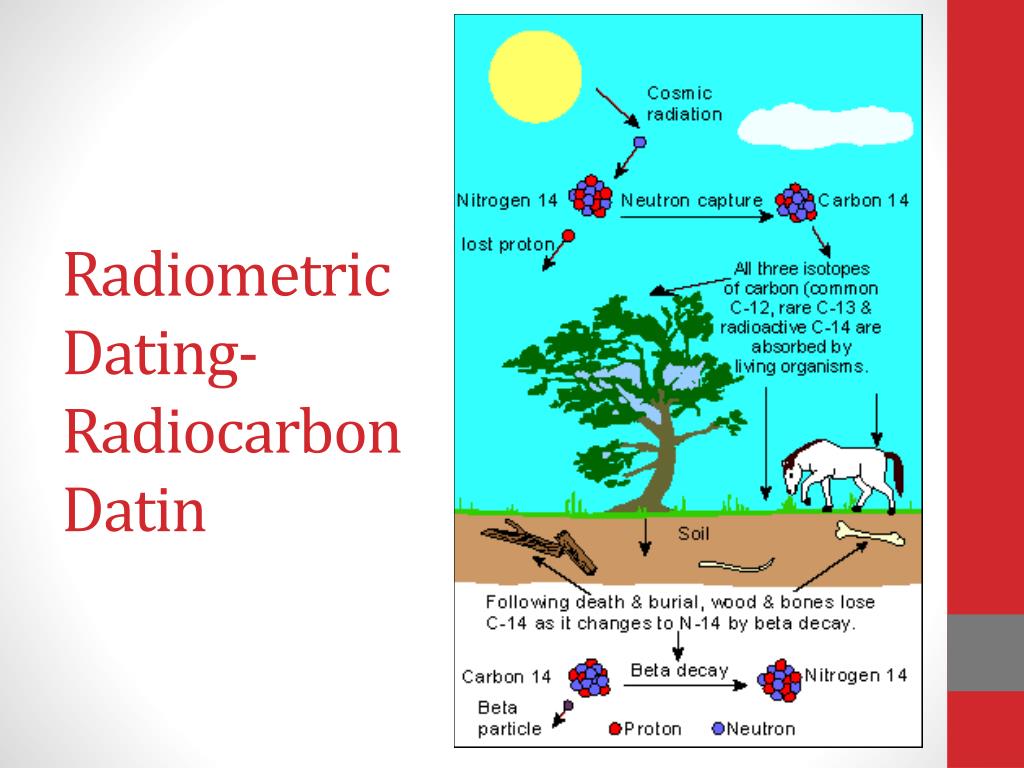
The length of time required for one-half of the unstable carbon –14 nuclei to decay (i.e., the half-life ) is 5,730 years. The difference between the concentration of carbon –14 in the material to be dated and the concentration in the atmosphere provides a basis for estimating the age of a specimen, given that the rate of decay of carbon –14 is well known. Whatever carbon –14 was present at the time of the organism's death begins to decay to nitrogen –14 by emitting radiation in a process known as beta decay. This carbon –14 cycles through an organism while it is alive, but once it dies, the organism accumulates no additional carbon –14. In any living organism, the relative concentration of carbon –14 is the same as it is in the atmosphere because of the interchange of this isotope between the organism and the air. Among the less abundant isotopes is carbon –14, which is produced in small quantities in the earth's atmosphere through interactions involving cosmic rays. Together carbon –12 and carbon –13 make up 99% of all naturally occurring carbon. The most abundant isotope in nature is carbon –12, followed in abundance by carbon –13. Carbon has isotopes with atomic weights between 9 and 15. He became intrigued by carbon –14, a radioactive isotope of carbon. The first method for dating organic objects (such as the remains of plants and animals) was developed by another American chemist, Willard Libby (1908 –1980). While the uranium-lead dating method was limited (being only applicable to samples containing uranium), it was proved to scientists that radioactive dating was both possible and reliable. Boltwood used this method, called radioactive dating, to obtain a very accurate measurement of the age of Earth. Thus, the greater the amount of lead, the older the rock. This was because uranium, as it underwent radioactive decay, would transmute into lead over a long span of time. In 1907, the American chemist Bertram Boltwood (1870 –1927) proposed that rocks containing radioactive uranium could be dated by measuring the amount of lead in the sample. However, many objects were found in caves, frozen in ice, or in other areas whose ages were not known in these cases, it was clear that a method for dating the actual object was necessary. By comparing the placement of objects with the age of the rock and silt layers in which they were found, scientists could usually make a general estimate of their age. It is based on the decay rate of the radioactive carbon isotope 14C, a form of carbon taken in by all living organisms while they are alive.īefore the twentieth century, determining the age of ancient fossils or artifacts was considered the job of paleontologists or paleontologists, not nuclear physicists. Carbon dating is a technique used to determine the approximate age of once-living materials. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
